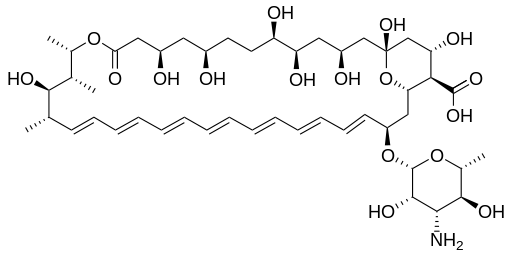
Polyene antifungal drugs for prion disease
Polyene antifungal drugs, most notably Amphotericin B (above) are a class of drugs which received some attention as potential anti-prion drugs in the 1980s and 1990s. The “-ene” suffix just refers to the many double carbon bonds in the molecule. Amphotericin B was isolated from Streptomyces bacteria in the 1950s and has long since passed into the public domain. As an antifungal drug, it binds to ergosterol, a small molecule found in fungal cell membranes (but not plants or animals), physically opening a small pore in the membrane which kills the cell by allowing interior and exterior ion concentrations to reach entropy.
Although we animals (and plants) don’t have ergosterol, we do have plenty of other sterols (such as cholesterol) in our membranes, and apparently polyenes aren’t totally specific, so they do have a tendency to tear holes in our membranes too [Laniado-Laborin 2009 (ft)]. They also appear provoke an immune response, destroy our kidneys and… the list goes on [ibid]. These are some really nasty molecules. They are still used intravenously sometimes, but only if a patient has an already life-threatening systemic fungal infection, since the drugs themselves are life-threatening.
These drugs also appear to have anti-prion effects. According to a review by Trevitt & Collinge 2006, the first to study them in animals was Amyx 1984, in a paper entitled “Chemotherapeutic trials in experimental slow virus diseases” in Neurology. This is such ancient history it’s not indexed by PubMed, and no copy (nor even Amyx’s first name) may be found online. But apparently Amyx tested Amphotericin B in rats, hamsters and even African green monkeys, and found a significant delay in onset. According to a later study [Pocchiari 1987 (ft)], Amyx’s rodent studies only looked at peripherally infected models, not at intracerebrally infected animals. But a startling number of animal studies were to follow, showing quite large delays in disease onset even in intracerebrally infected animals.
All of the studies that examined Amphotericin B’s efficacy reported significant extensions of survival [Pocchiari 1987 (ft), Pocchiari 1989, Xi 1992, McKenzie 1994], though just as you’d expect, the effect was heavily dependent on both the dose and how early in the disease course the drug was administered. The experiments where the animals started receiving the drug on the same day they were infected showed delays of 50% or even up to 87%, while when the drug wasn’t administered until 28 days post infection (dpi) the delay was just a statistically insignificant 3%. In all of these studies, just one experiment [in Pocchiari 1987 (ft)] waited until the animals became symptomatic and then administered the drug, and found no effect. Most of the experiments used a dose of 1 mg/kg/day 6 days per week (that’s in hamsters, so equivalent to about 8 mg/day for a 70 kg person) or 2.5 mg/kg, but the experiments that got up to 10 mg/kg found many of the mice dying of drug toxicity before they died of scrapie infection.
As mentioned earlier, Amphotericin B’s existing clinical use is largely limited to patients with life-threatening systemic fungal infections. It’s relatively hard to get a systemic fungal infection if you have a functioning immune system. Many of the patients receiving AmB in the 1980s and 1990s were AIDS patients, and the search for less toxic alternatives for treating these patients led Cefai 1991 to report on the development of MS-8209, a new AmB derivative with similar efficacy that was about five times less toxic.
A second wave of prion-infected animal studies compared MS-8209 with AmB [Demaimay 1994, Adjou 1995, Adjou 1996, Demaimay 1997, Adjou 1999, Adjou 2000]. These generally found that MS-8209 was considerably less toxic than AmB and could be administered at higher and more effective doses – delays in death of up to 100% were reported. One troubling finding among these studies was that both of these drugs’ efficacy appeared to be strain-specific. Adjou 1996 reported that the drugs could delay death in mice by 10-15% when the mice were infected with C506M3 prions (a rodent strain of scrapie) but only a barely significant ~5% when the mice were infected with bovine spongiform encephalopathy prions (BSE, i.e. the mad cow strain of prions, but passaged in mice).
To my knowledge, the only other polyene ever tested in prion-infected animals was mepartricin, which Pocchiari 1989 found to be ineffective. At least one other polyene made it into some cell culture experiments. Marella 2002 (ft) reported on the efficacy and possible mechanism of action of filipin.
Upon looking up the dosage for Amphotericin B I see that drugs.com has a pretty wide range of possible doses depending on the indication. Many of the indications call for 0.25mg/kg/day, so that’s 17.5 mg/day for a 70 kg person. Doses are sometimes escalated to as much as 1 mg/kg/day – so potentially 70 mg/day. The 70 kg human equivalents of the animal doses used in all of these studies were generally in the 8 – 20 mg/day range for Amphotericin B, and a bit higher for some MS-8209 experiments due to its lower toxicity.
When I review studies of potential anti-prion drugs I usually try to include a table summarizing the animal survival or delay-in-onset data. Extracting all these data from the papers was an unfathomably tedious process. I count 85 separate experiments across 10 studies, varying almost every possible aspect of the experiment: dose, animal type, prion strain, infection method, when the treatment was started, and when it was stopped. The one thing that appears to have been constant is the use of intraperitoneal injection as the treatment route – since polyenes are BBB-permeable, no one felt the need to try intracerebral injection. Here’s my best attempt at summarizing the data in some sort of remotely intelligible table:
It’s really staggering: the polyenes turn out to be by far the best-studied class of antiprion drugs, at least in terms of animal survival studies. Just look at the repeated names on these papers: it strikes me that K.T. Adjou and R. Demaimay (both in the Dormont lab at Universite Denis Diderot in Paris) must have put their entire postdoctoral years into studying these drugs.
All that, and then everyone just dropped the subject after 2000 and apparently never looked back. Even Adjou 2000 concludes by touting the likely efficacy of these drugs – no one ever seems to have concluded, publicly at least, that the drugs didn’t deserve further study.
Do they deserve further study? They certainly wouldn’t be at the top of my list. The sole report in the literature of their use in humans [Masullo 1992] reports no therapeutic effect, in two symptomatic CJD patients treated with AmB escalating to 1 mg/kg/day. That’s wholly consistent with Pocchiari 1987 (ft)’s finding that AmB didn’t delay ‘advanced clinical signs’ when hamsters were treated beginning at the onset of ‘clinical signs.’ sCJD patients (and plenty of genetic prion disease patients too) are not diagnosed until symptoms are present, perhaps even advanced, at which point no evidence suggests that polyenes would be helpful. The only humans who could undergo presymptomatic treatment, equivalent to the early-treated mice and hamsters listed above, would be carriers of genetic prion diseases, but the toxicity of the polyenes (and the fact that MS-8209 never became an FDA-approved drug) makes them a pretty unappealing preventative option.
These drugs, then, suffer from the same practical paradox as intrathecally infused pentosan polysulfate: the only people they might benefit are not desperate enough to take the risk.
That said, I am not prepared to entirely forget about this class of drugs. An effective treatment may ultimately require a cocktail of several drugs, and given the wealth of evidence that AmB and MS-8209 do act to inhibit prion propagation and slow disease, they could yet be part of a solution.