ASOs at single cell resolution
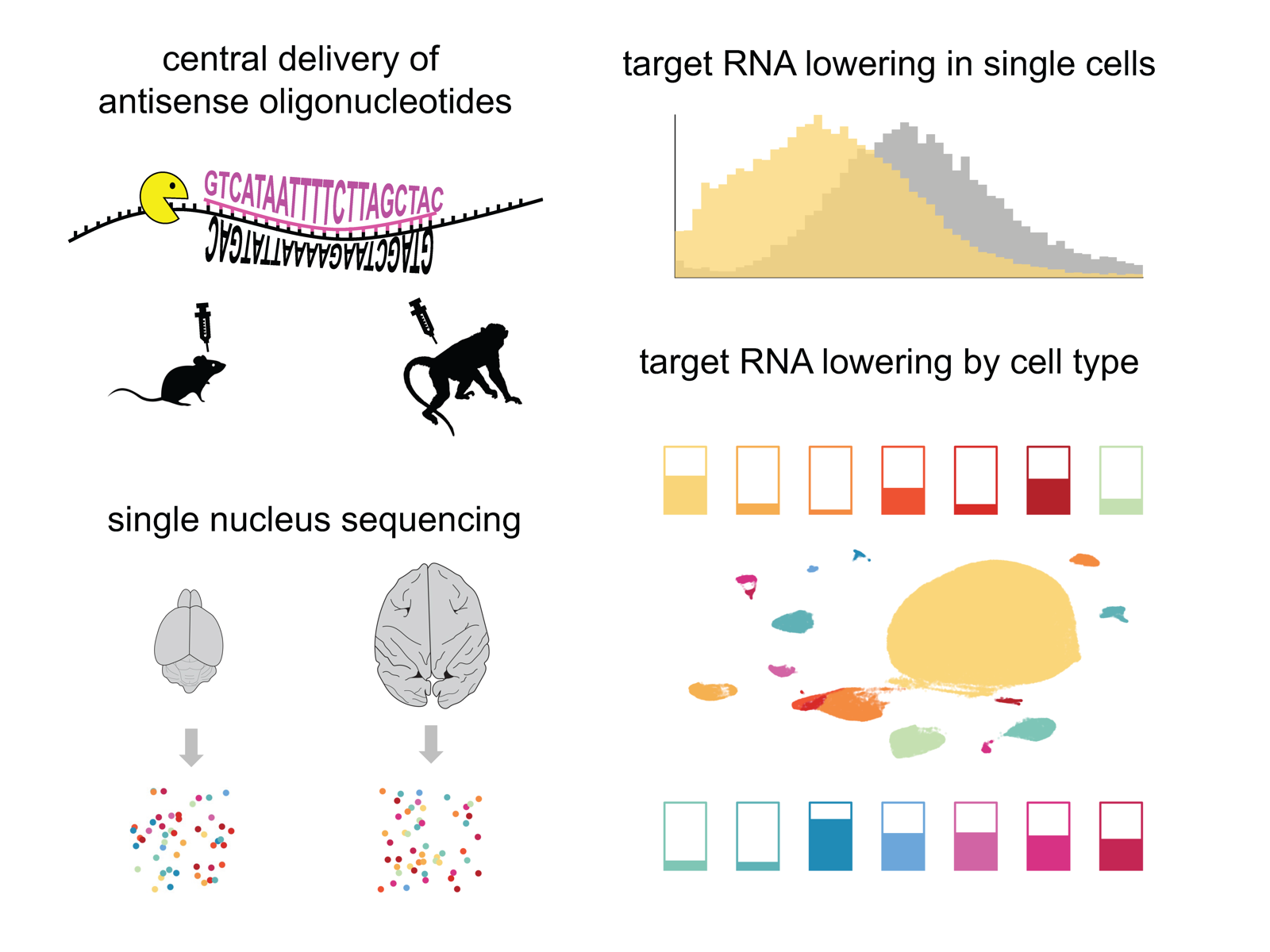
Today marks the official publication of our study of antisense oligonucleotide (ASO) activity in the brain using single nucleus sequencing [Mortberg & Gentile 2023] (full text). I unpacked many of the key findings in a tweetstorm when the preprint first came out, and many folks in the oligonucleotide medicine community will have seen me present this work at Cold Spring Harbor in March. With the final publication now out, I want to also take a few moments to explain how the work came about, its potential significance for the prion community.
As I’ve blogged previously, we really want to prevent prion disease — not just treat after it strikes — and this will require a new strategy in clinical trials. Therefore in 2017 we began meeting with FDA scientists to discuss a potential clinical path for primary prevention in prion disease. Our proposal has been to test a PrP-lowering drug in people at genetic risk for prion disease, and use PrP concentration in cerebrospinal fluid (CSF) as a primary endpoint of the trial. If the drug lowers CSF PrP, we believe that could be a basis for Accelerated Approval. FDA scientists agreed with the rationale for prevention in prion disease, and with the idea that PrP is so biologically central to prion disease that measurement of PrP could serve as a primary endpoint. But that’s not a stamp of approval — instead FDA gave us a lot of homework! They told us that many aspects of our data package including the biomarker and the animal studies will need to be extremely strong to support such a novel pathway, and they asked us to do a lot more work to shore up the arguments for our strategy.
This new paper responds to one piece of homework they gave us. One was to make sure that the biomarker will actually reflect the cell types we care about. Prion disease only kills neurons, and neuronal PrP knockout is sufficient to confer protection from neurotoxicity. Apart from neurons, lowering PrP might help only a little in some cell types (astrocytes), or be totally irrelevant in others (microglia, oligodendrocytes). Could our biomarker ever lie to us? What if an ASO lowered CSF PrP, and we clapped ourselves on the back and said “we did it!” only to realize years later that we’d only lowered PrP in glia and not done the job in neurons. Therefore, as the meeting minutes from our first 2017 encounter record, “FDA recommended the conduct of studies to assess the distribution of PrP in different cell types in the brain, and how effective ASOs are in reducing PrP expression in these different cell types.”
Sonia and I transformed that mandate into a funding proposal to Ono Pharma Foundation, whose Oligonucleotide Medicine Award seemed a perfect fit. We would treat animals with ASOs in a number of different paradigms, and quantify target RNA lowering in all different cell types. In our interview, David Corey, the chair of the Scientific Advisory Board, grilled us on how committed we really were to the project, and whether it was truly critical path. After surviving the interview by what we thought was the skin of our teeth, Dr. Corey revealed that he actually loved the proposal, he just wanted to make sure the project wouldn’t be a distraction from our core mission. To our delight, Ono Pharma Foundation funded the three-year proposal in 2019.
The work was hard! Meredith Mortberg spent the better part of a year shadowing Naeem Nadaf and Chuck Vanderburg from the Macosko lab to learn how to do the cryostat dissection, purify nuclei, and get high-quality sequencing data out of it. She eventually settled on a 9-hour hands-on protocol with only an awkwardly timed 15-minute break at 10:45 am for lunch. Juliana Gentile took a deep dive on identifying markers and assigning cell types to the data, and checking whether the RNA lowering from bulk tissue matched what we got from single-nucleus sequencing. Hien Zhao and Holly Kordasiewicz from Ionis provided tissue, and used their knowledge of ASO pharmacology and biology to help us identify the right questions to ask. Sean Simmons brainstormed with me to devise the statistical models. It seemed like a huge team and three years seemed like a long time, but in the end we needed everyone, and every minute of that time, and then some.
We hope that the data and the methods established here will be broadly useful to people working on ASOs and other oligonucleotide medicines, such as siRNAs. The dataset and source code are publicly available on GitHub, and if you need even raw-er data, it’s on Broad’s single cell portal.
But for the prion community, there are really two key findings worth highlighting. These points are relevant to Ionis Pharmaceuticals’ ASO program in prion disease, which hit some delays last year but remains active, with prospects of a first-in-human clinical trial hovering in the near future.
First: ASOs lower target RNA in just about every single cell in the brain. An ages-old question — Sonia and I get asked this in the Q&A of almost every talk we give — has been whether 50% knockdown means you lowered the target by 50% in 100% of cells, or by 100% in 50% of cells. The answer is actually somewhere in between, because we found that different cell types do have some differences in degree of target RNA reduction. But it’s a bit closer to the former, in that ASOs do appear to lower the target RNA at least somewhat in 100% of cells, at least in brain regions that take up a reasonable amount of drug. But note that this is just the answer for ASOs! Different drugs could have very different answers. Another question we always get in Q&A is “why can’t we just solve this problem with CRISPR”. The short answer to that is: drug delivery. The biggest (though not only) problem is it’s still really hard to deliver CRISPR systems, or any other gene therapy, to every neuron in the brain. Imagine a future where people finally succeed in delivering such large cargoes to the human brain. Then it might become possible, by targeting the gene at the DNA level, where there are just 2 copies, to complete silence/edit/delete the gene in the cells you do reach, while achieving nothing at all in the cells you don’t reach. This will be a distinction for everyone interested in neurological drugs to keep their eyes on in the coming years. Two drugs could look similar at the level of a biomarker or a bulk tissue, but one could be half-rescuing all cells, while one could be completely-rescuing half of cells. I’m not taking a position on which option is better: in fact, the best option is often going to be both. But for some diseases and targets, the difference between half-in-all and all-in-half could be important, and is worth keeping in mind.
Second: neurons track well with overall target RNA lowering. We found that ASOs are active very broadly across cell types in the brain, including all different subtypes of neurons, as well as everything else. Granted, there were meaningful differences in degree in some cases. For instance, when an ASO lowered a target RNA to 45% of its original level in the cerebellum, that corresponded to 58% in granule neurons but 17% in Purkinje neurons. But for PRNP, a target where we believe that less is always better, the evidence suggests that the overall bulk signal will never gravely mislead you. CSF PrP is presumably some kind of average of all different brain regions and multiple cell types, and since it’s just one value, it’s never going to perfectly reflect everything you care about. But across every experiment we did, spanning different animals, ASOs, targets, and brain regions, we never found a single neuronal subtype where the relative PrP level was more than 11% higher than the bulk tissue. Indeed, when we saw that CSF PrP was lowered to 60% of its original level, every type of neuron had PrP RNA lowered by at least that much, often considerably more.
Treating healthy pre-symptomatic people at risk for genetic prion disease is a big undertaking. For the plan to work, a lot of things will all have to line up: we’ll need to be able to identify and recruit a high-risk population, the drug will have to be quite safe, the biomarker will have to be quantitative, there will need to be plans for follow-up and monitoring of people, and so on. A lot of what our lab has done for the past six years, and is continuing to do, is to try to lay every piece of this foundation for a primary prevention strategy to stand on. Our brains depend on every single pillar being sturdy. This new study helps to shore up one more pillar. If an ASO is able to lower PrP in CSF in at-risk people, the new findings give us confidence that this will indeed mean that we lowered the problem-causing protein in the cells we care most about.
